Make better development decisions - Earlier
Partner with us to bring Clarity, Confidence, and Speed to your drug development programs.
3
15+
Years of experience
Countries
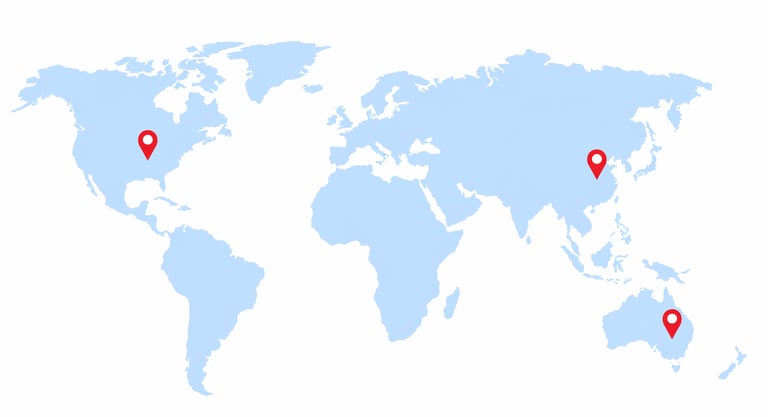
China: Shanghai & Chengdu
Australia: Sydney & Melbourne
USA: California
Fu Shuai, PhD
President, Principal Consultant
Dr. Fu is a senior expert in quantitative pharmacology and Bayesian statistics, with more than 10 years of cross-disciplinary global experience in model-driven drug development and clinical value assessment. Her expertise spans PK/PD modeling, Bayesian decision frameworks, model-based meta-analysis (MBMA), network meta-analysis (NMA), and real-world data analytics, with a strong focus on translating quantitative evidence into clinical and strategic decisions.
Ellen Zhang
Chief Commercial Officer
Ms. Zhang brings over 20 years of experience in pharmaceutical and life sciences industries, specializing in end-to-end solution building across the full drug lifecycle, from early development to commercialization. Her leadership has been recognized with multiple industry honors, including Top Performer in Business Development and President’s Club Member awards.
A seasoned R&D leader with over 20 years of global experience and expertise in Oncology, Ophthalmology, CNS, and Rare Diseases. Possesses end-to-end drug development expertise, including strategic portfolio planning, Phase I–IV clinical trial management, and global and regional regulatory filings.
Wang Qiao, MSc
Medical Director
Our Team








Kenny Chen
Senior Modeling &
Analytics Director MD
A pharmacometrics leader with over 10 years of experience in data analysis and Model-Informed Drug Development (MIDD). Expert in population PK, PBPK analysis, and complex model development and validation. Proven track record leading modeling strategies for global programs from early research through pivotal trials and regulatory submissions, with strong expertise in anti-infectives and other innovative therapeutic areas.


Jason Zhao, PhD
Quantitative Pharmacologist &
Computational Biologist
A pharmacometrics expert with over 10 years of experience in PK/PD modeling and systems pharmacology across small molecules and biologics. Specialized in Model-Informed Drug Development (MIDD), including NLME, mechanistic PBPK/QSP models, and multi-omics data–driven modeling. Applies Machine Learning and AI to support lead selection, trial design, exposure–efficacy–toxicity prediction, and model-driven dose optimization for complex clinical programs.


Wenping Wang
Principal Consultant
Dr. Wenping Wang is an internationally recognized leader in MIDD and pharmacology, with over 20 years of deep industry experience spanning in pharmaceutical consulting and innovative drug development. He has held senior scientific roles at, including Pharsight (Certara), Johnson & Johnson, Novartis, and Takeda, and has contributed to the successful approval of multiples NDAs across diverse therapeutic areas including oncology, cardiovascular, central nervous system, and other disease areas.
量科达医药科技(成都)有限公司 | 统一社会信用代码:91510100MACRQWYQIN
© 2026. All rights reserved.
